|
However, the ever increasing knowledge about the complexity of molecular regulatory networks derived from numerous experiments on knockout animals, functional and high throughput studies based on tissue homogenates (genome and proteome sequencing) puts additional demands on IF/IHC-based research of human tissue samples 1. Following the advent of antibodies targeted against specific proteins (and to a certain degree other classes of molecules such as glycans), IF is being regularly applied as a complementary method for the molecular profiling of tissue samples, which in turn is useful for the diagnostic purposes (subtyping of diseases such as tumors, inflammatory diseases, autoimmune disorders), and for the evaluation of outcomes of different experimental procedures performed in basic biomedical research. 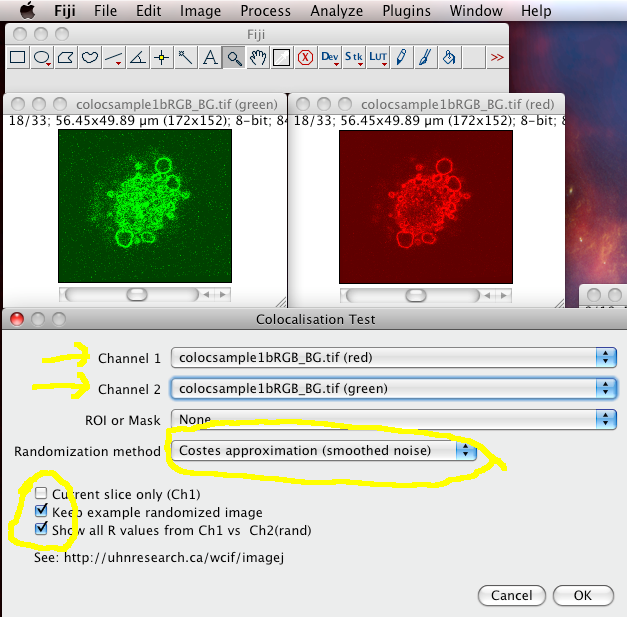
The immunofluorescence (IF) (and immunohistochemistry (IHC) in general) has long been recognized as one of the fundamental methods for biomedical research. Combined with the data from ontological bases, this approach to quantification of IF enables creation of in silico virtual disease models. This can serve as an alternative for colocalization of IF staining of multiple primary antibodies based on repeating cycles of staining of the same histological section since those techniques require non standard staining protocols and sophisticated equipment that can be out of reach for small laboratories in academic settings. Although the standard IF staining protocol is applied, the data output enables colocalization of multiple markers (up to 30) from a given histological sample. There is no selection of multiple Regions-Of-Interest (ROIs) since the entire histological section is quantified. No cell counting is applied either to determine the cellular content of a given histological section nor the number of cells positive to the primary antibody of interest. This approach to quantification of IF staining utilizes pixel (px) counts and comparison of px grey value (GV) or luminance. 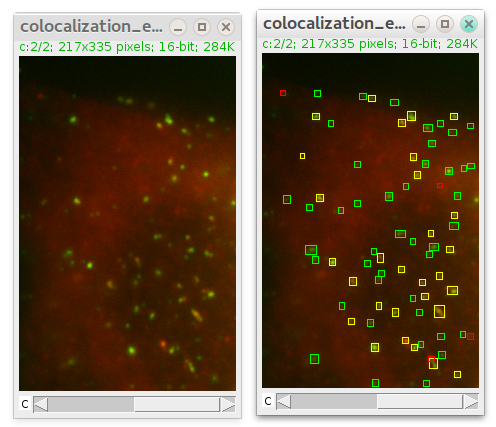
The importance of histomorphometric profiling of tissue samples and IF signal thresholding is elaborated. Expression domains and spatial gradients of IF signals were quantified by histograms and 2D plot profiles, respectively. Human gingiva samples stained with primary antibodies against the common leukocyte antigen CD45 and factors related to heparan sulfate glycosaminoglycans (HS GAG) were used. We describe a novel approach for quantification and colocalization of immunofluorescence (IF) signals of multiple markers on high-resolution panoramic images of serial histological sections utilizing standard staining techniques and readily available software for image processing and analysis.
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
